
It is also a complete Chemical Equations Solver. It also comprises all chemistry formulas and equations to solve and provide you the right answer by measuring equation problems. Remember that the Chemical Equation Balance App does not contain only the facility of equation balance. Quick results of the chemistry balancer. One second for balancing the Chemical Reaction. Balancing chemicals calculator for all. You can also copy and paste equations or results of your balanced equation practice by solving Chemistry Equations and formulas with this calculator. Then this Chemical Equation Solver process on your query, solve equation and Reactions to show accurate results in no time. You only need to write your desired equation and tap the balance button. This Chemical Equation Balance App enables you to solve equations in only a few seconds. We sure it will be your next favourite app as Chemical Balancer. So that, you’ll be able to find the Chemical Formulas and Chemistry Equations in no time by using this Chemical Equation Balancer and chem calculator. Good thing of this chemistry equation app is it also contains a periodic table. This app contains all chemistry equations, so you can easily measure and balance equation of chemistry with this Chemical Equation Balancing Calculator. Students of any level and class can use this chemistry solver app because we have designed it with an easy to use interface for balancing chemical equation. This Chemical Equation Balance App is a smart tool which works accurately as a chemical balancer. It gives you the finest experience to solve problems of equations and chemical reactions with a chemistry calculator. Multiply the number of calcium atoms by 3 and the number of phosphorus atoms by 2.Chemical Equation Balancer - Chemistry calculator to measure formulas with Chemical Equation Balancer.Ĭhemical Equation Balancer to easily measure chemistry formulas. Phosphorus goes from 0 to -3, gaining 3 electrons (oxidation).Calcium goes from 0 to +2, losing 2 electrons (reduction).Identify the changes in oxidation numbers:.In Ca 3P 2, calcium has an oxidation number of +2, and phosphorus has an oxidation number of -3.Phosphorus (P) also has an oxidation number of 0 in its elemental form.Calcium (Ca) has an oxidation number of 0 in its elemental form. 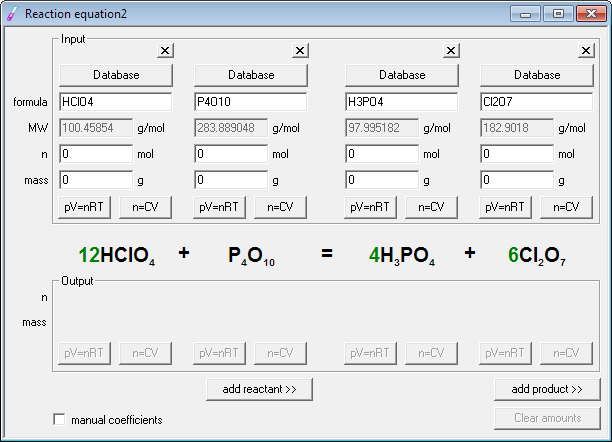
Process: identify the oxidation numbers, determine the changes in oxidation state, balance the atoms that change their oxidation state, and then balance the remaining atoms and charges. Useful for redox reactions, this method involves balancing the equation based on the change in oxidation numbers.īest For: Redox reactions where electron transfer occurs. Balance the oxygen atoms by placing a coefficient of 2 in front of H 2O:.There are 2 O atoms on the left and 1 O atom on the right. There are 2 H atoms on the left and 2 H atom on the right. Count the number of H and O atoms on both sides.Process: Start with the most complex molecule or the one with the most elements, and adjust the coefficients of the reactants and products until the equation is balanced. It involves looking at the equation and adjusting the coefficients to get the same number of each type of atom on both sides of the equation.īest for: Simple equations with a small number of atoms. Balancing with inspection or trial and error method A balanced equation obeys the Law of Conservation of Mass, which states that matter is neither created nor destroyed in a chemical reaction. However, this equation isn't balanced because the number of atoms for each element is not the same on both sides of the equation. For example, in the reaction of hydrogen (H₂) with oxygen (O₂) to form water (H₂O), the chemical equation is: It shows the reactants (substances that start a reaction) and products (substances formed by the reaction). calcium hydroxide + carbon dioxide = calcium carbonate + waterĮxamples of the chemical equations reagents (a complete equation will be suggested):Ī chemical equation represents a chemical reaction.PhCH 3 + KMnO 4 + H 2SO 4 = PhCOOH + K 2SO 4 + MnSO 4 + H 2O. 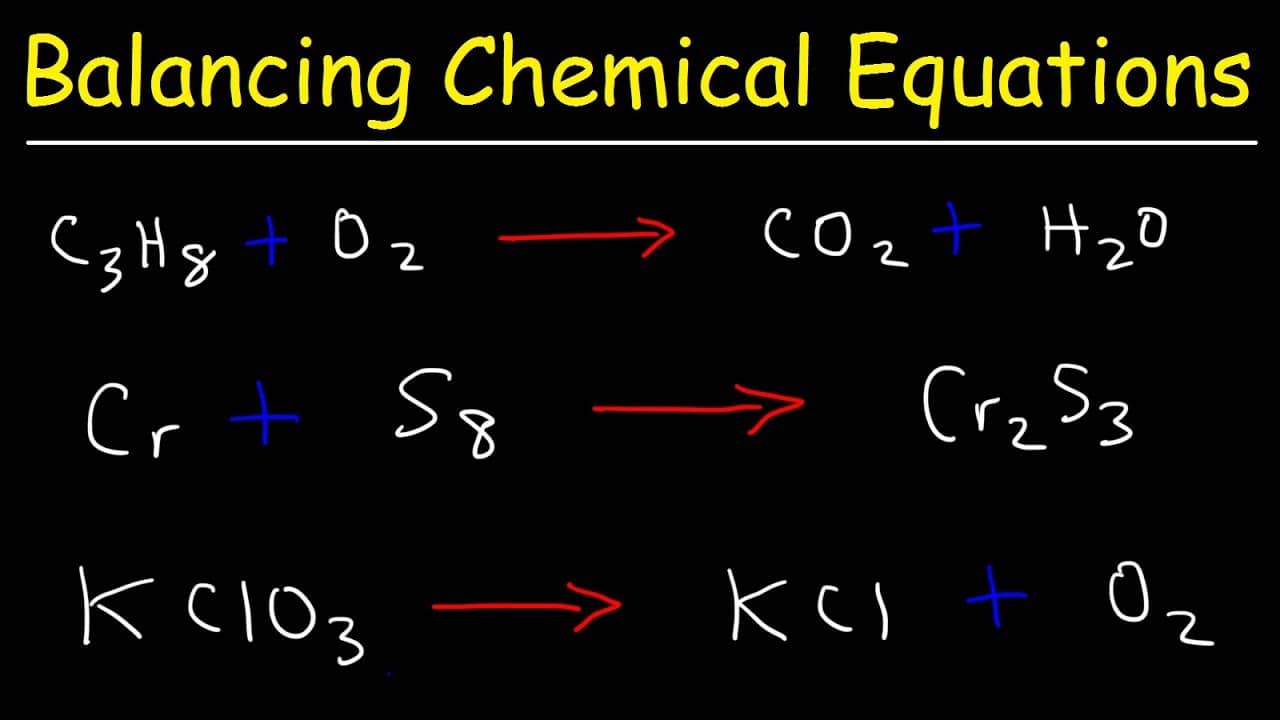
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
